 IV Anesthesia - Barbiturates
IV Anesthesia - Barbiturates
Barbiturate Development
Basic Barbiturate Pharmacology
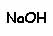
Chemistry and Formulation
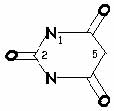 <-->
<-->
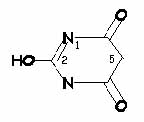
keto-enol tautomerism of barbituric acid,
devoid of hypnotic activity
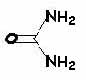
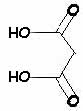
Barbituric acid may be described as a "cyclic ureide of malonic acid:"
 +
+
 ->
->

 +
+
 ->
->
 +
+
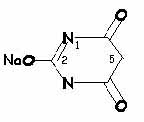
Hypnotically active barbiturates:
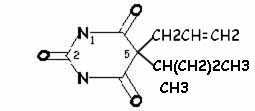 |
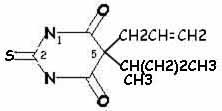 |
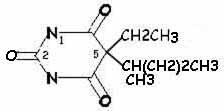
| secobarbital | thiamylal |
 |
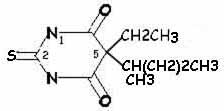 |
| pentobarbital | thiopental |
 |
|
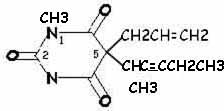
| methohexital | |
Na salts + 6% by weight NaCO3 + H2O or NS
Buffering action of NaCO3 plus atmospheric CO2 maintains pH at 10 to 11. In less alkaline solutions, these barbiturates may precipitate as the free acids; so do not reconstitute with NS or LR; and do not mix with acidic solutions of other drugs. Properly reconstituted, refrigerated solutions of thiobarbiturates are stable for 1 week, while similar solutions of methohexital are stable for 6 weeks.
Structure-Activity Relationships
Hypnotic activity: side chains at position 5 (especially if one of them is branched)Potency and duration of action: length of side chain at position 5 (so, secobarbital and thiamylal are slightly more potent than pentobarbital and thiopental, respectively)
More rapid onset and shorter duration of action: sulfur instead of oxygen atom at position 2 (so thiamylal and thiopental have more rapid onset and shorter duration of action than secobarbital and pentobarbital, respectively)
Increased incidence of excitatory side effects: methylation at position 1 (methohexital)
Increased potency, rate of onset and short action: increased lipophilicity
Stereoisomerism Though their l-isomers are nearly twice as potent as their d-isomers, barbiturates are marketed as racemic mixtures. Methohexital has two asymmetric carbon atoms, so exists as 4 stereoisomers (alpha,beta-d,l-methohexital). The beta isomers are associated with extensive motor activity, so methohexital is marketed as racemic alpha-d,l-methohexital. Different activities among different stereoisomers (enantiomers or enantiomorphs) is consistent with a site of action at a chiral center of a receptor or enzyme.
Mechanism of Action
Most likely site of action: gamma-aminobutyric acid (GABA) receptor complex, GABAA.Effects on GABAA occur at clinical drug concentrations, correlate with anesthetic potency and are stereospecific.
GABA is the principal inhibitory neurotransmitter in the mammalian CNS
GABAA complex:
- oligomeric complex of 4 to 6 glycoprotein subunits assembled to form a ligand-gated chloride ion channel
- activation (e.g. by GABA) leads to increased chloride conductance causing hyperpolarization, hence inhibition or decreased excitability, of the postsynaptic neuron
Barbiturates enhance and mimic the action of GABA at the GABAA receptor complex. Barbiturate binding to this receptor decreases the rate of GABA dissociation and increases the duration of GABA-activated chloride channel opening. At slightly higher concentrations, barbiturates directly activate chloride channel opening even in the absence of GABA, leading to "barbiturate anesthesia."
Pharmacokinetics
An intravenous barbiturate bolus distributes first into a "central blood pool" chiefly perfusing relatively low-volume, high blood flow organs such as the brain. (Despite high affinity, adipose tissue takes up drug slowly due to relatively low perfusion.) Elimination clearance contributes almost nothing to termination of induction effect. Drug effect is terminated in large part by uptake into lean tissues such as muscle. First-pass pulmonary uptake of thiopental is about 14%.| Mean data from compartmental models | |||||
|---|---|---|---|---|---|
| VC (L/kg) | Vdss(L) | ClE(ml/min/kg) | t1/2beta(hrs) | Hepatic ER | |
| thiopental | 0.38 | 2.5 | 3.4 | 11.6 | 0.15 |
| methohexital | 0.35 | 2.3 | 10.9 | 3.9 | 0.50 |
Pharmacodynamics
Pharmacodynamics may be most easily elucidated by studying a continuous noninvasive measure of drug effect: for thipental, the electroencephalogram (EEG). Thiopental causes a biphasic EEG response with activation at low concentrations followed by inhibition at higher concentrations. However, a unique relationship between thiopental's EEG effect and anesthetic depth (measured by response to noxious stimuli) could not be established.Clinical Pharmacology and Uses
Altered responses to induction doses of barbiturates are due altered pharmacodynamics or early distribution pharmacokinetics. The classic example is the markedly reduced dose required in the presence of hypovolemic shock. In this situation, the brain receives a higher fraction of the induction dose and removal is quite slow because of decreased blood flow to other organs. The decreasing dose requirement with age is probably related to the relative decrease in lean body mass with age and the relative increase in adipose tissue. For similar reasons, induction dose is decreased in females and in the obese.It has been suggested that thiopental dose(mg) = 350 + wt - 2(age) - 50(if female)
Induction of General Anesthesia
Barbiturates induce general anesthesia rapidly and pleasantly (painlessly). They act in one arm-to-brain circulation time with maximum effect in about 1 minute and duration about 5-8 minutes. Induction doses produce the highest blood concentration, the greatest effects on body systems and the most side effects.Usual, recommended induction doses of thiopental:
| adults | 2.5-4.5 mg/kg |
| children | 5-6 mg/kg |
| infants | 7-8 mg/kg |
Premedication and ASA Physical Status influence induction dose (mg/kg):
| Premed | ASA I | ASA II | ASA III & IV
| none
| 4.0 |
3.7 |
3.2 |
benzodiazepine
| 3.4 |
3.3 |
3.1 |
light opioid
| 3.4 |
3.1 |
3.0 |
heavy opioid
| 3.1 |
2.8 |
2.7 |
|
|---|
Methohexital is about 2.7 times more potent than thiopental. Methohexital 1.5 mg/kg is approximately equivalent to 4.0 mg/kg thiopental.
Thiamylal is not significantly different from thiopental in potency, incidence of laryngospasm, respiratory depression, cardiotoxicity or recovery time.
Injection complications
- urticarial rash
- upper chest, neck, face
- fades in a few minutes
- probably due to histamine
- upper chest, neck, face
- anaphylactoid reactions are occasionally seen
- hives, facial edema, bronchospasm, shock
- absence of reaction to oral barbiturates does NOT ensure lack of sensitivity to IV barbiturates
- treatment symptomatic: may include epinephrine in 100 mcg increments, IV fluids, bronchodilators
- hives, facial edema, bronchospasm, shock
- pain on intravenous injection - uncommon: thiopental 1-2% and methohexital <5% when injected into small veins
- intra-arterial injection or subcutaneous extravasation
- methohexital -> mild discomfort, no sequelae
- thiopental extravasation
pain edema, erythema -> sequelae ranging from slight soreness to extensive tissue necrosis depending on concentration and total amount injected
- thiopental intra-arterial
may cause intense arterial spasm with possibly severe pain at and distal to the injection site which may persist for hours and be associated with anesthesia or hyperesthesia of the distal extremity, edema or motor weakness
sequelae range from mild discomfort to gangrene and loss of distal tissue
Treatment is aimed at preventing permanent sequelae and consists of
- diluting the drug and relieving spasm: papaverine 40-80 mg in 10-20 ml NS intra-arterial or
- lidocaine 1% 5-10 ml intra-arterial, and
- consider sympathetic block (stellate ganglion block or brachial plexus block) to relieve spasm, and
- heparin IV to prevent thrombosis
Specific Organ Function Effects
CNS Effects
- Barbiturates may be hyperalgesic in subanesthetic doses.
- Thiopental produces dose-related depression of the EEG:
- delta and theta waves (increasing amplitude and decreasing frequency)
- burst supression
- flat line
- Thiopental 4 mg/kg/hr maintains flat EEG (as may pentobarbital infusion that maintains a plasma concentration of 3-6 mg%).
- Thiopental causes a dose-related decrease in CMRO2 to 55% with flat EEG reflecting a decrease in neuronal but not metabolic need for O2. (Hypothermia is the only way to decrease the metabolic O2 requirement.)
- Thiopental decreases CBF and ICP. CPP is maintained because ICP decreases more than arterial pressure.
- Methohexital is associated with seizures after high doses; and 33% incidence of postoperative seizures when given by continuous infusion.
- SSEP's remain even after doses of thiopental that flatten the EEG, but median nerve SSEP and BSAEP responses change in a dose-dependent fashion.
Intraocular Pressure
IOP decreases 40% after an induction dose of thiopental or methohexital. If succinylcholine is given immediately after thiopental, IOP returns to preinduction value. (Delay in giving succinylcholine leads to more elevated IOP.)Respiratory Effects
- central respiratory depression
- both rate and depth of breathing are decreased
- respiration apparently returns toward normal in a few minutes, but responses to hypercarbia and hypoxia remain depressed for a longer time
- low incidence of hypersalivation
- rare bronchospasm
- laryngeal reflexes are more active (intact) after thiopental than after propofol
- mucociliary clearance depressed (to same degree as with the volatile agents)
Cardiovascular Effects
- predominant one: venodilation leading to peripheral blood pooling
- myocardial contractility is depressed to a lesser extent than with volatile agents
- baroreflex mechanism only slightly depressed
- SVR usually unchanged (decreases 21% in patients with preload-independent artificial hearts and constant cardiac output)
- thiopental also dilates pulmonary vessels
- methohexital: heart rate increases more than with thiopental
- no arrhythmias occur (unless hypercarbia or hypoxia occur)
- decreased sympathetic output from the CNS
- do not sensitize the heart to catecholamines
- use with caution whenever increase in heart rate (especially methohexital) or decrease in preload may be detrimental to the patient (e.g.: pericardial tamponade, hypovolemia, CHF, ischemic heart disease, heart block)
